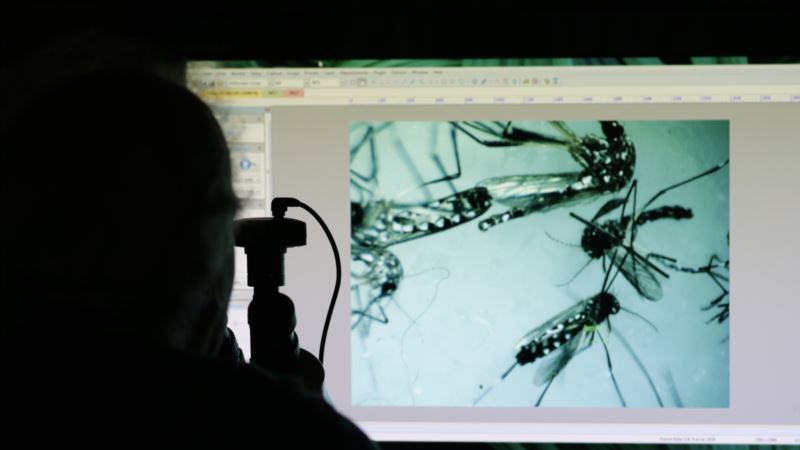
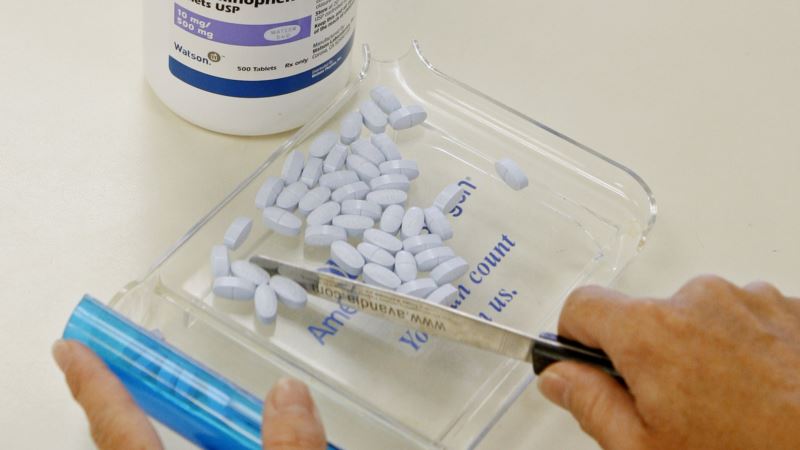
Researchers in the United States have identified a number of drug compounds that stop the spread of the Zika virus, potentially protecting pregnant women and their unborn babies from the mosquito-borne illness. One of the compounds already is used to treat tapeworms. Researchers at the U.S. National Institutes of Health screened a library of 6,000 existing drugs and experimental compounds. Then, investigators at Florida State University in Tallahassee and Johns Hopkins University in Baltimore tested the drugs. The compounds were shown to block the ability of the Zika virus to kill cells and to keep the microbe from reproducing within infected cells. Both classes of compounds were neuro-protective, showing activity in fetal brain cells affected by microcephaly. They include neural progenitor cells that contribute to the development of the fetal brain. Scientists found that the compounds work either before or after exposure to Zika, and that they are most effective when more than one is used at the same time. Discovery of the compounds was published in the journal Nature Medicine. Power of suppression Hengli Tang, Florida State University Biological Sciences professor and researcher, said that if and when the drugs become available, they would be used to thwart transmission and would not be used in pregnant women. "But even if you can treat Zika-infected men or non-pregnant women, you can still have the … potential to cut down transmission,” Tang said, “because with the suppression of viral replication, you reduce the amount of virus that's in an infected person and that can reduce the chance of transmission by mosquito bites or sexually." Earlier this year, Tang's team was the first to show how Zika caused microcephaly. The search is urgent for a drug to treat Zika or to prevent transmission of the virus, which can cause microcephaly in developing fetuses. Babies with microcephaly are born with abnormally small heads and brains, causing a host of disabilities. The disease already has infected thousands of pregnant women from Brazil through North America. In people who are not pregnant, the virus causes relatively minor flu-like symptoms, but also can cause a rare neurological disease in adults called Guillain-Barre syndrome. Hope in ‘repurposed’ drugs Because at least one of the promising drugs already is approved by U.S. regulators, doctors could prescribe now, though it is not known how much of the compound should be given or how often. Tang said it is possible researchers eventually could formulate the drugs to give to pregnant women. "If a compound is modified and the anti-viral is modified so it's safe to use in pregnancy,” Tang said, “then the mechanism of the anti-viral would be to reduce the virus to a low enough level that it will have a much lower or zero chance of getting to the fetus to begin with. And that's sort of the idea of an anti-viral." The compounds have been what scientists called "repurposed," meaning they already have received regulatory approval for use or testing in humans for other disorders. That could speed the time it takes to get Zika drugs to the public. Even so, Tang predicts it might be two more years before that happens.
Researchers Find Drug Compounds to Target Zika